Genera analyses merger option
Genera Biosystems (GBI) announced this week (June 30, 2014) that it has identified its preferred transaction from the strategic review process it began during 2013.
We wrote on GBI with a speculative “Buy” recommendation in Genera waits for ideal partner on June 11.
| The company is currently loss-making with minimal revenues, and is largely dependent on a partner to take the next step to profitability. Therefore, the stock is clearly high risk, and our “Buy” recommendation is Speculative. |
Details of potential merger
Although the company involved in the merger proposal has not been named, the preferred transaction involves a merger with a larger international molecular diagnostic business with a highly compatible platform technology that is achieving considerable commercial success while growing rapidly.
It is claimed the partner is a larger international molecular diagnostics company and has a number of key strategic relationships that can be leveraged creating substantial revenue opportunities for the combined group. These relationships include a large pharmaceutical business,and substantial leading US pathology customers.
Further, the partner company has well-developed relationships with certain global IVD (in vitro diagnostics) companies that are also preferred IVD partners for Genera.
Our view
The announcement clearly reads very positively and would suggest a significant re-rating when all terms are finalised and a binding merger implementation agreement is executed.
However it needs to be cautioned that, without any final details on the structure of the merger and who the partner is, GBI remains very speculative with high risk.
There is the risk of continued delays. Management had previously stated on numerous occasions that the outcome from its strategic review would be completed by June 30. It is now saying the final details of the Merger Implementation Agreement won’t be announced for another six weeks.
GBI is suggesting this is an interim step to a partnering deal with a global IVD company or a takeover. It is important that this initial partner already has revenues, and the combined group will be able to increase revenues at a faster rate than GBI could do by itself.
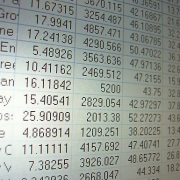
The large global IVD companies have a history of paying approximately 6-10 times revenue when they take strategic stakes via partnering deals or acquire companies outright, which the table below outlines. Note that the lower multiple paid for Innogenetics N.V. was driven by the fact that approximately 46% of its revenues were derived by lower margin non-molecular diagnostic products.
Further, these large IVD companies look to outright acquisitions of targets that generally are delivering revenues of greater than US$20 million. As assumed in our previous note we believe that GBI can generate approximately $13 million in revenues in 2016 alone and given the recent announcement we assume that the merger partner may deliver revenues larger than GBI’s in 2016. If the initial merged group could get to $20-40 million revenue, then the share price upside in a final sale to a global IVD company is large – especially as GBI’s market cap is currently only $30 million.
Health Canada approves Roche’s HPV test
Since our initial GBI recommendation, the other material news that has been released is the Canadian approval of Roche’s HPV test as a primary cervical cancer screening tool. This follows the US FDA approval of Roche’s HPV test in April this year.
Alongside the Australian government recommendation of HPV testing as the primary screening tool, a global trend appears to be developing. We would expect other countries to follow suit, with similar announcements from European countries especially.
One of the Roche’s tests key advantages is it ability to simultaneously genotype two of the high-risk strands of HPV. These two types are responsible for about 70% of cervical cancer.
The reason Roche’s approval is material for GBI is that GBI’s test has been independently assessed as more clinically advanced. That is, it can simultaneously genotype all 14 high risk types of HPV, which are responsible for 99.7% of cervical cancer cases.
The independent assessment was done by the Wolfson Institute in London, and included a comparison against the other commercially available HPV tests.
The reason it is important to have the ability to individually genotype the HPV is because it improves the efficiency and accuracy of the screening process. For example, just because a type of HPV has been identified does not necessarily place a female in the high-risk category for cervical cancer. However, if the same high-risk type of HPV is found over multiple years, then it places the female in the high-risk category and the patient can be directed to the next stage of testing. The next phase of testing involves costly and invasive tests such as colposcopy/biopsy.
Whilst HPV tests have previously been used in combination with pap smear tests, the global trend of approving HPV tests as a stand-alone cervical cancer screening tool is obviously a positive development.
We are not suggesting the HPV test will completely replace the pap smear, but the proven clinical advantages of HPV tests such as GBI’s over pap smears should ensure increased global demand for GBI’s “PapType HPV” technology. As a guide the recent Australian move to adopt HPV testing as the primary screening tool ahead of pap smear in 2016 will result in an approximately 90% decline in pap smear volumes from their current level of 2.25 million per annum.
Summary
Although GBI appears to be heading in the right direction, the high-risk nature of our recommendation can’t be overstated. A successful merger would de-risk the stock and enable greater visibility towards the company’s value creation opportunity.
We maintain our “Speculative Buy” recommendation with a price target of $0.54.
Simon Dumaresq owns shares in Genera Biosystems.